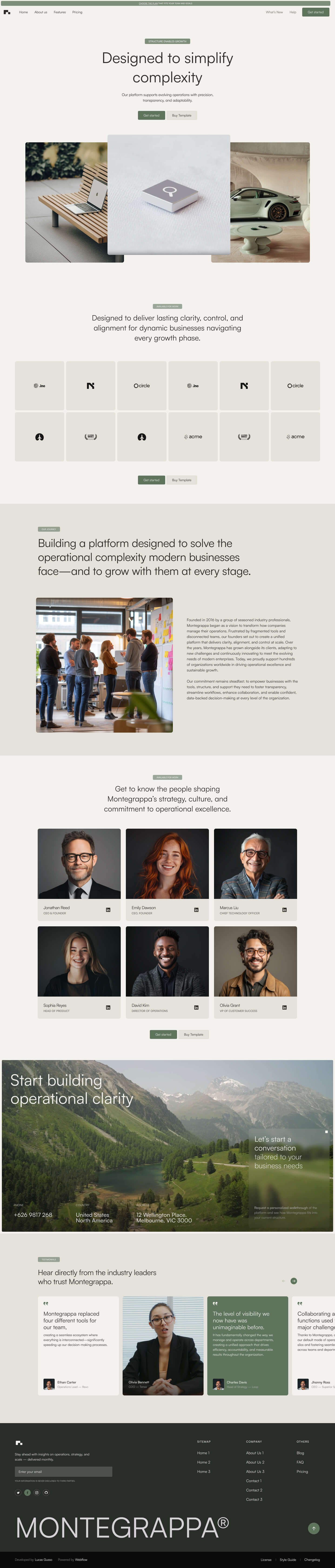
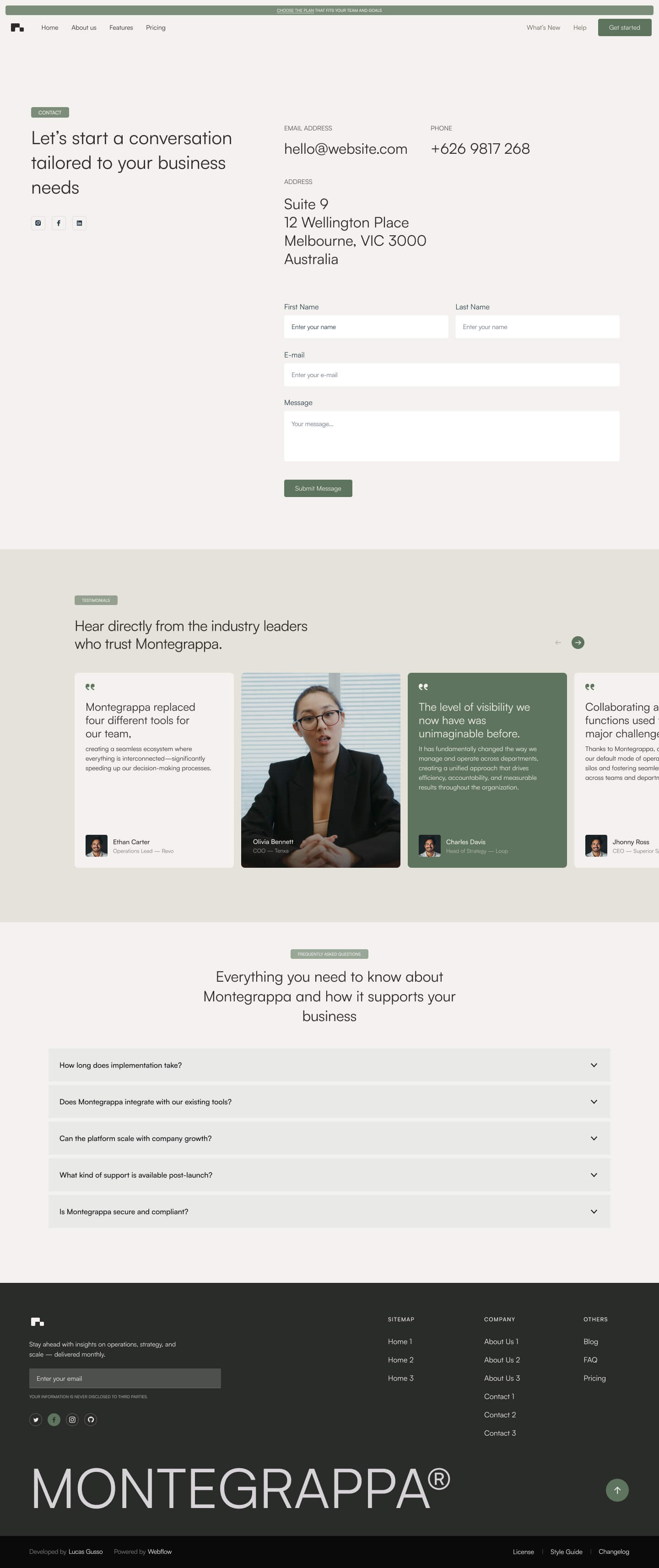
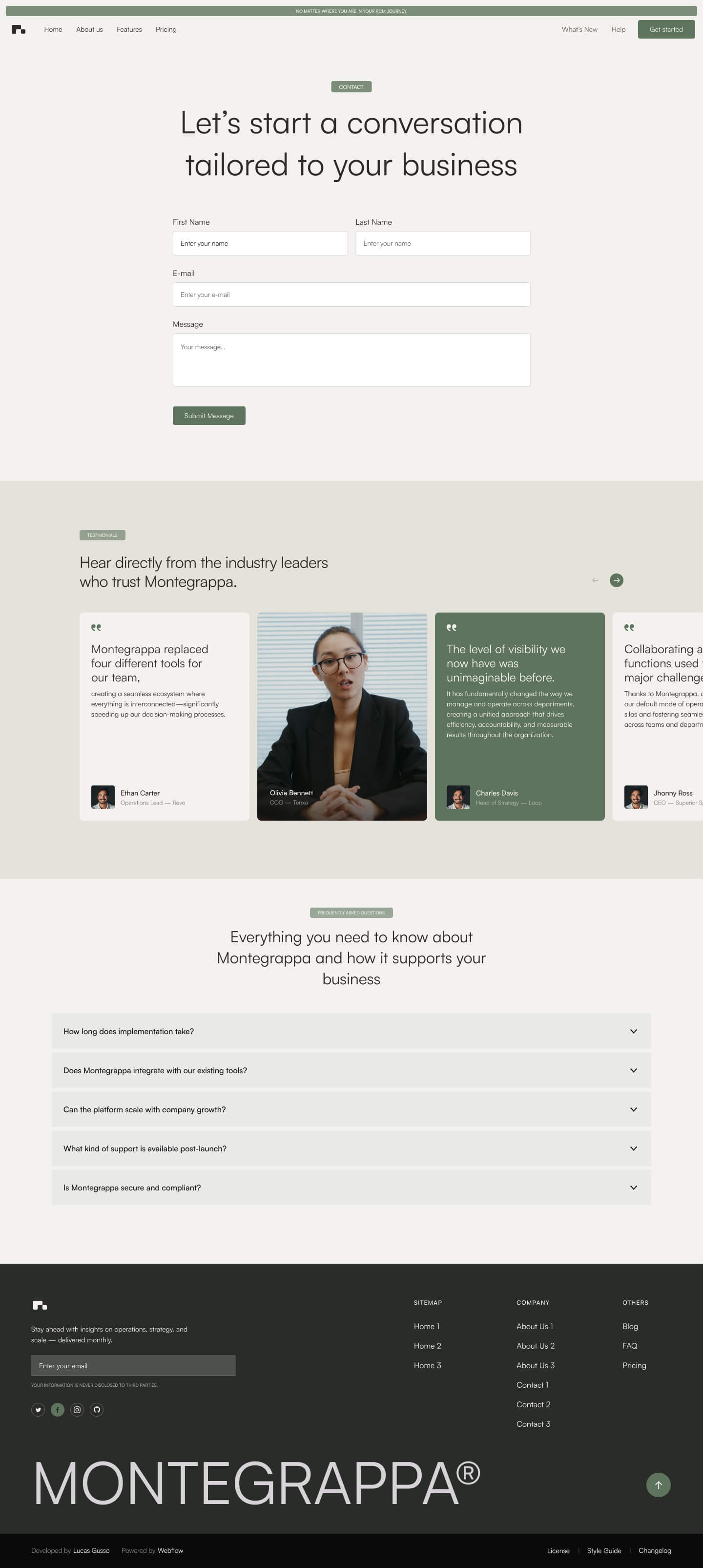
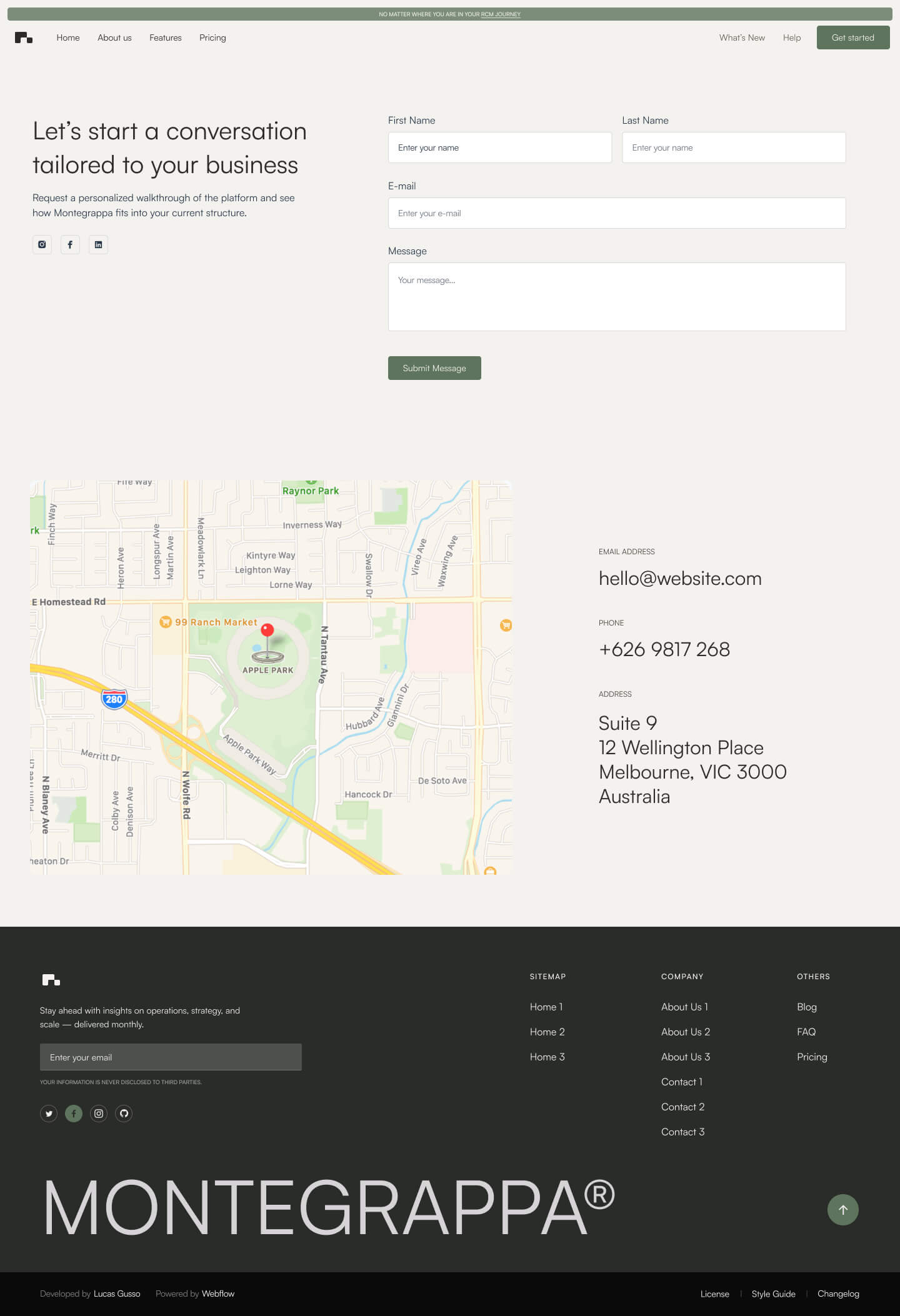
Get a closer look at every layout included in the Montegrappa Template. Each homepage variation is designed for clarity, flexibility, and performance—so you can choose the one that best fits your needs.
Send us an email to lucas.gusso@gmail.com with your purchase receipt, and we will send you the editable Figma file for the Montegrappa Template.
The First State-Regulated Psilocybin Study for Veteran PTSD in U.S. History.
One veteran at a time.
No matter where you we expected performance improvements, but the strategic implementation delivered unprecedented operational excellence of delivering products.
Every day, over 44 veterans die by suicide in the United States. Not as a statistic, as fathers, mothers, daughters, sons. People who served with extraordinary courage and came home carrying wounds that conventional medicine was never fully able to heal.
.png)
Treatment-resistant PTSD is not a failure of willpower. It is a failure of tools. Emerging research from Johns Hopkins, NYU, and Imperial College London has demonstrated that psilocybin-assisted therapy produces profound, lasting relief, in cases where decades of conventional treatment had failed.
What has been missing is the rigorous, real-world evidence base needed to bring these treatments to national policy. That is precisely what this study is designed to produce.
Veterans with treatment-resistant PTSD
Psilocybin sessions per participant
Oral psilocybin dose, FDA Breakthrough Therapy
Oregon and Colorado's regulated frameworks
The Protocol
Half a Century of Research.A Question No One Has Answered.
Every one of those trials was conducted in a sterile clinical setting. None has answered the critical question: do these results hold inside a real-world, state-regulated therapeutic system? As the only research institution embedded within Oregon and Colorado's regulated programs from their inception, CIRC is uniquely positioned to answer it, and to generate the evidence that policy requires.
Out of 870 individuals assessed for eligibility in a landmark randomized clinical trial, only 27 met the criteria to enroll — illustrating the scientific rigor these studies demand.
.png)
Figure 1. CONSORT Diagram of Participant Flow. Source: Davis AK, Barrett FS, May DG, et al. Effects of Psilocybin-Assisted Therapy on Major Depressive Disorder: A Randomized Clinical Trial. JAMA Psychiatry. 2021;78(5):481–489. doi:10.1001/jamapsychiatry.2020.3285. CIRC's veteran PTSD study follows the same rigorous screening standards.
A Structured Path to Federal Impact
Protocol Development: Team assembled. Site agreements in place.
Fundraising Close + IRB: $650K goal reached. IRB submitted.
IRB Approval + Recruitment: Veteran recruitment opens.
Onboarding + Preparation: 30 veterans selected and prepared.
Study Launch: Dosing sessions begin across both states.
Integration + Data: Follow-up, PCL-5 assessments, full dataset.
Results Published: Peer-reviewed findings submitted and published.
Federal Policy Impact: DEA conversations. Multi-site follow-up.
This Study Doesn't End With 30 Veterans. It Begins There.

The data generated here will travel far beyond Oregon and Colorado. It will contribute the first state-regulated, real-world psilocybin PTSD dataset to the federal evidence base, the kind of evidence that moves DEA scheduling conversations, shapes FDA policy, and builds the case for a national standard of care.
Once validated, the protocol becomes replicable across every state-regulated program in the country. What begins here becomes the evidence that a generation of policymakers has been waiting for.
When you fund this study, you are not writing a check to a research lab. You are writing the first chapter of a new system of care.
Every Dollar Has a Job.
Facilitator time, sessions, healing center fees, and psilocybin for all 30 veterans.
Principal investigator, research coordinators, IRB compliance, and two years of study administration.


$0 raised of $650,000 goal
Machine learning processing, peer-reviewed publication, and open-access distribution to policymakers and clinicians.